 “There has been nothing for these patients that has adequately addressed their severe AA symptoms, and baricitinib looks to be a game changer in the treatment and management of patients suffering from this psychologically devastating disease. “The approval of baricitinib for severe AA means that patients with this awful disease may now finally get treatment,” said Brett King, MD, PhD, associate professor of dermatology at Yale School of Medicine in New Haven, Connecticut. Baricitinib, a JAK inhibitor, modulates the activity of these 2 cytokines, leading to hair regrowth in AA. AA is mediated by cytokines interferon-γ and IL-15, which signal through the JAK-STAT pathway. Once an adequate response is achieved, the daily dose can be reduced to 2 mg, giving clinicians and their patients flexibility in disease management. ”Clinicians can titer up or down based on the patient’s hair loss severity and individual needs. This is the first time that patients with severe AA will get an FDA-approved oral systemic agent demonstrating excellent efficacy in the treatment and management of their disease. “Similarly, 1 in 3 adults can achieve 80% coverage with a 4-mg daily dose of baricitinib. 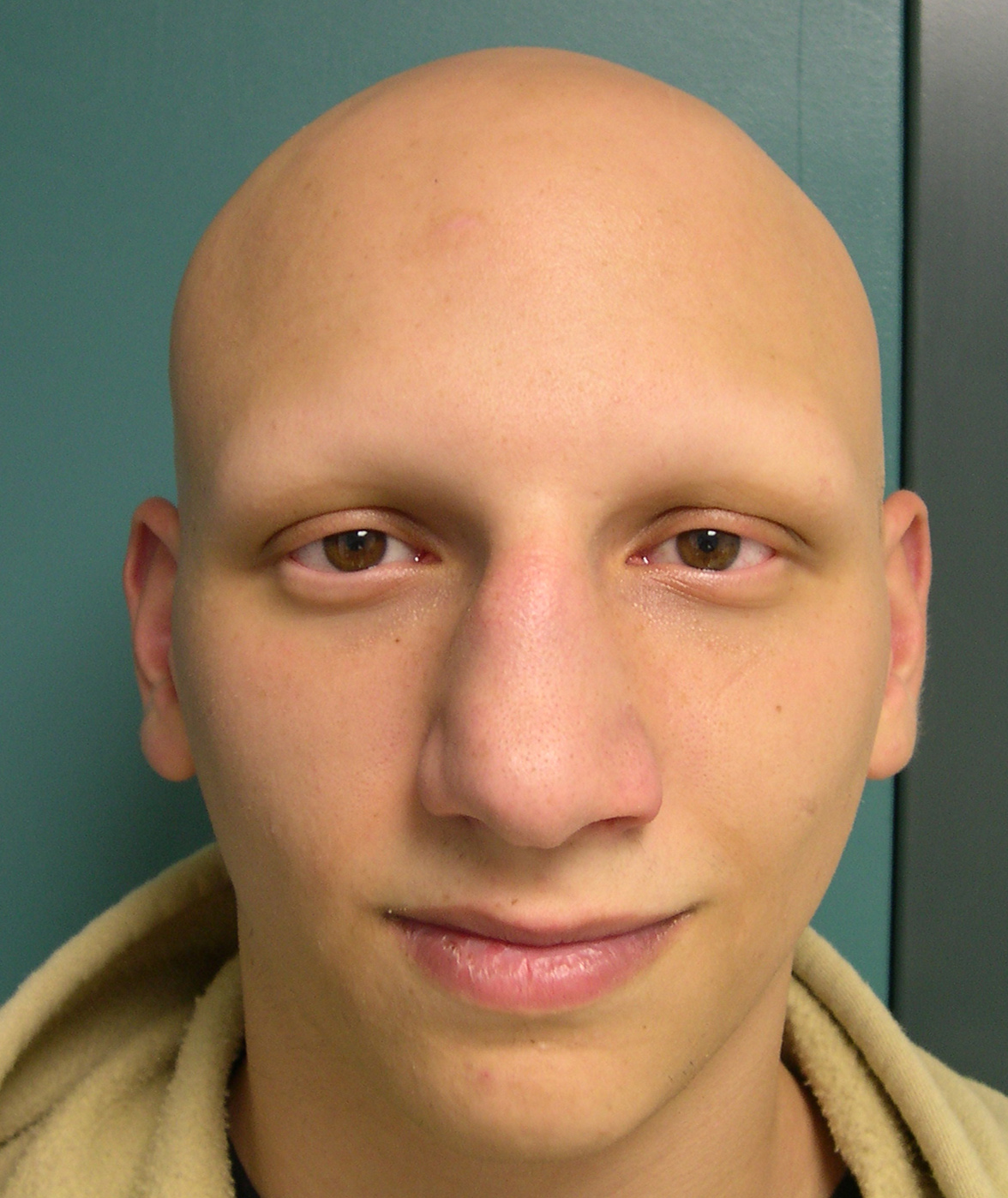
“We can freely say that 1 in 5 adults with a 2-mg daily dose of baricitinib can achieve 80% of regrowth of hair coverage,” Mallbris said. In BRAVE-AA2, 17% of the 156 patients who received 2 mg of baricitinib and 32% of the 234 patients who received 4 mg of baricitinib achieved adequate scalp hair coverage vs 3% of the 156 patients who received a placebo. Results from BRAVE-AA1 showed that 22% of the 184 patients who received 2 mg of baricitinib and 35% of the 281 patients who received 4 mg of baricitinib achieved adequate scalp hair coverage compared with 5% of the 189 patients who received a placebo. 
The primary end point of the trials was the proportion of patients who achieved at least 80% scalp hair coverage at week 36. Study participants were placed into 1 of 3 arms and received placebo, 2 mg of baricitinib, or 4 mg of baricitinib every day. ”The data behind the historic FDA approval for severe AA comes from 2 trials 4, BRAVE-AA1 (NCT03570749) 5 and BRAVE-AA2 (NCT03899259) 6, which were randomized, double-blind, placebo-controlled studies including patients presenting with at least 50% scalp hair loss as measured by the Severity of Alopecia Tool for more than 6 months. Baricitinib is a huge therapeutic breakthrough for this patient population. This can be deeply traumatic for patients, and they deserve a safe and effective treatment for their disease. “These patients not only have patchy hair loss but can also lose their eyebrows and eyelashes, and when this happens, they cannot recognize themselves in the mirror anymore. “Unfortunately, AA is sometimes wrongfully characterized as a cosmetic disease,” said Lotus Mallbris, MD, PhD, vice president of global immunology development and medical affairs at Eli Lilly and Company in Chicago, Illinois. This can result in significant anxiety and depression and can have a devastatingly negative psychosocial impact, particularly when symptoms are severe-further impacting a patient’s quality of life. The clinical presentations for AA can vary from 1 to multiple well-defined patches to more diffuse or total hair loss on the scalp (alopecia totalis) or hair loss on the entire body (alopecia universalis). 
“This approval will help fulfill a significant unmet need for patients with severe alopecia areata.”ĪA, which affects approximately 6.8 million people in the United States, 2is a common autoimmune disorder with a still-unknown etiology typically resulting in unpredictable patchy hair loss on the body. “Access to safe and effective treatment options is crucial for the significant number of Americans affected by severe alopecia,” said Kendall Marcus, MD, director of the Division of Dermatology and Dentistry in the FDA’s Center for Drug Evaluation and Research, Washington, DC, in the FDA announcement. In June 2022, the FDA ended the wait for a specific treatment for this condition with its approval of the novel oral J Kinase (JAK) inhibitor baricitinib (Olumiant Eli Lilly and Company and Incyte) as the first systemic treatment specifically indicated for severe AA. Alopecia areata (AA) is a historically challenging-to-treat condition, with few effective therapy options. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
